 
To overcome this limitation of UV/Vis spectroscopy for atomic analysis we must devise an instrument that is invariant to the matrix of the elements.Not very useful (we say it is not very robust) See Robustness and Ruggedness at chemlibree.Result is that any calibration curve that you create will only work for a very narrow, limited sample matrix.Molecules : A molecule is an electrically neutral group of two or more atoms held together by chemical bonds. So this implies changing the matrix in a solution will change the spectrum of the analyte. Examples of elements are Hydrogen, Carbon, Oxygen, Nitrogen etc.This process is explain by Crystal field theory (inorganic class) chemlibre page.Each spectrum holds a wide variety of information. Since unique elements have unique emission. Figure 10.7. Spectra can be produced for any energy of light, from low-energy radio waves to very high-energy gamma rays. In physics, atomic spectroscopy is the study of the electromagnetic radiation absorbed and emitted by atoms. (a) A sample of excited hydrogen atoms emits a characteristic red light. the amount of d splitting of the transition metal Cr varies by the strength of the electric field and its geometry (square planar, tetrahedral, octahedral, etc.) created by the electrons in the ligands. Atomic emission occurs when a valence electron in a higher energy atomic orbital returns to a lower energy atomic orbital. For example, because hydrogen is the most basic Atom, it has the most basic Spectrum. These so called line spectra are characteristic of the atomic composition of the gas. 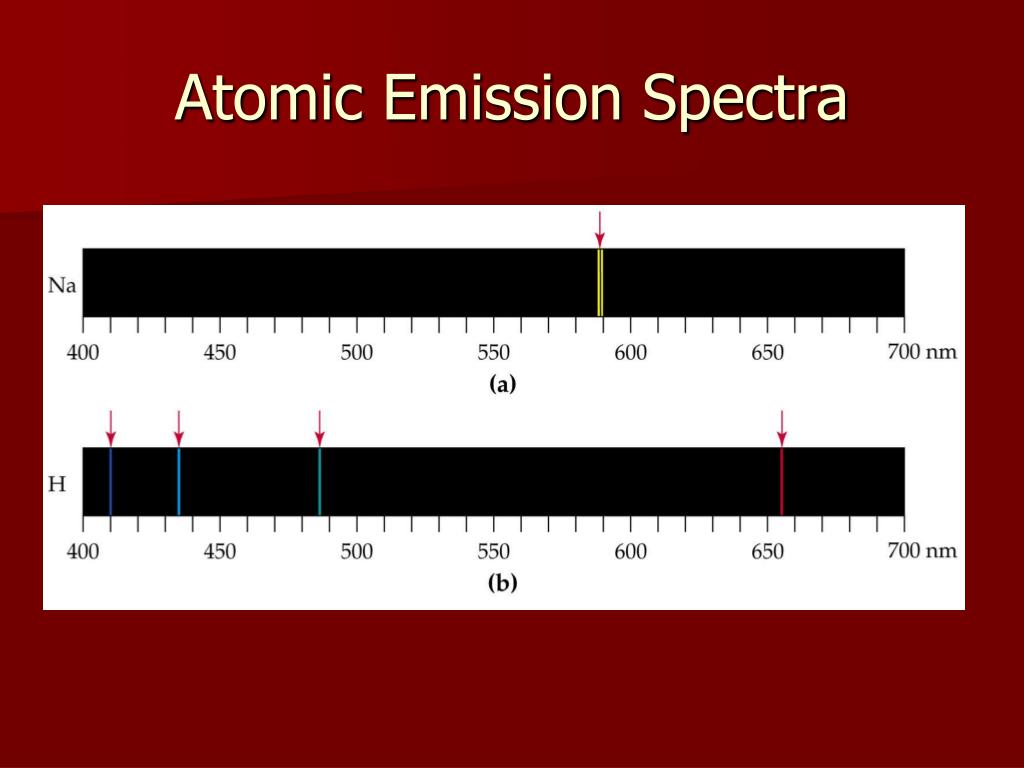
the d orbitals are split by ligands bound to the metal (called d-orbial splitting) What is the energy of the photon emitted when a Hydrogen atom makes a transition from the displaystyle n4 to the.the transition being measured by UV/Vis spectroscopy is a d → d transition.What does this figure imply about using UV/Vis spectroscopy for determining the concentrations of metals or even the presence the element. Everyday examples abound: the stove element in the kitchen, the metal filament in a lightbulb, and even the Sun.Figure 1: UV/Vis spectrum of Chromium cations with 2 different ligands (NO 3 and Cl) in water and ethanol 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
